Translate this page into:
Combined approach with therapeutic drug monitoring and pharmacogenomics in renal transplant recipients
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
In patients undergoing renal transplantation, dose individualization for tacrolimus is routinely achieved with therapeutic drug monitoring (TDM). The patient started on 5.5 mg/day of tacrolimus had a significantly elevated tacrolimus trough concentration. The tacrolimus dose was regularly reduced following TDM at many time periods in the post transplant period but the tacrolimus concentration was consistently elevated. Genomic analysis done after four years revealed mutations in the genes encoding for CYP3A5 and MDR1 (2677G > T). Pharmacogenomics alongside TDM, will soon emerge as the backbone of dose individualization. But for genomics to be beneficial, it should be advocated in the pre-transplant or early post transplant period.
Keywords
Drug monitoring
polymorphism
tacrolimus
Introduction
Tacrolimus-based regimen that includes induction with daclizumab and maintenance with mycophenolate mofetil and corticosteroids has superior efficacy in renal transplantation when compared with cyclosporine-based regimens.[12] Pharmacodynamic and pharmacokinetic variability with tacrolimus have been attributed to various factors such as co-medication, biochemical, hematological parameters, and genetic constitution.[34] Dose individualization for tacrolimus is routinely achieved with therapeutic drug monitoring. Recent literature highlights personalizing drug therapy using the pharmacogenomic approach.[5]
Inter-individual variability of tacrolimus is attributed to polymorphisms of Cyp3A5 and transporters as P glycoprotein.[67] The presence of Cyp3A5*3 allele, CYP3A5*6, or MDR-1 (T-1236C, C3435T, and G2677T) are associated with an increased exposure to tacrolimus. Scholten et al. reported that tacrolimus area under the concentration time curve (AUC0-12) of 210 ng.h/mL for 6 weeks post-transplant and 125 ng.h/mL thereafter would be adequate to prevent acute rejections.[8] For mycophenolate, the dosing is based on maintaining AUC0-12 within 30-60 mg.h/L.[9]
Case Report
A 50-year-old lady of mongoloid extraction from Bhutan, with chronic kidney disease resulting from membranous proliferative glomerulonephritis pre-emptively received a renal allograft from her haplo-matched cousin sister. Her pre-transplant donor-recipient crossmatch and donor-specific antibodies were negative. She received standard immunosuppressive therapy with mycophenolate sodium (1440 mg/day), tacrolimus (5.5 mg/day), and steroid (20 mg/day) with basiliximab induction. She was also prescribed valganciclovir, cotrimoxazole, pantoprazole, and calcium. During the first 2 months post-transplant the MPA AUC0-12 ranged from 29.2 to 168.7 mg.h/L with total daily doses varying from 1440 to 1620 mg. She developed leukopenia which did not remit with reduction in dose of mycophenolate. Due to persistent leucopenia, other drugs such as valganciclovir and co-trimoxazole were stopped. Mycophenolate dose was gradually tapered but leucopenia continued to worsen therefore mycophenolate was stopped. Subsequently, leucocyte counts never increased more than 5000/cu.mm so mycophenolate could not be restarted. Renal and liver function parameters were within normal limits in the post-transplant period. The tacrolimus trough measurements (ng/ml) and the subsequent dose adjustments over time are shown in Figure 1.
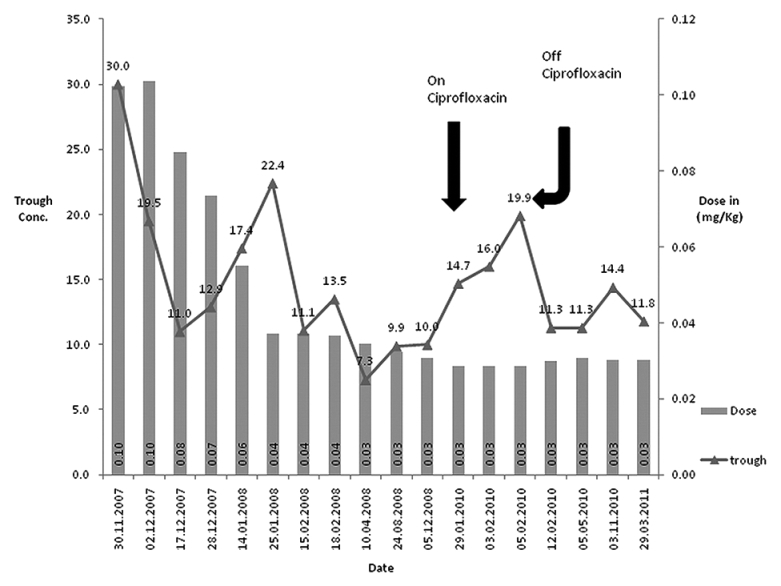
- Tacrolimus dose (mg/kg) and trough measurements (ng/mL) over time
Dose reductions for tacrolimus were made gradually over a 1½ month period. However, the trough concentration remained above the recommended trough concentration. Tacrolimus 2 point LSS AUC within the first post-transplant year ranged between 165.6 and 140.9 μg.h/L while on 2 mg/day in two divided doses, which was within the range of exposure in the maintenance period at this center.[10] Post-transplant immune monitoring with donor-specific antibody was negative throughout her follow-up. BK virus PCR screening remained negative when tested during serial visits.
Two years later, the two-point LSS tacrolimus AUC increased to 281.7 μg.h/L after initiation of ciprofloxacin which reduced to 217.3 μg.h/L, 1 week after discontinuation of ciprofloxacin.
In view of the possibility of polymorphism affecting tacrolimus metabolism, with informed consent, single nucleotide polymorphisms that could account for the elevated tacrolimus levels were tested for with PCR. She was mutant homozygous for CYP3A5*3(GG) alleles and MDR1-2677(TT) alleles, heterozygous for MDR1-1236(CT) alleles and wild-type homozygous for CYP3A5*6 (GG) and MDR1 3435C (CC) alleles.
Discussion
The optimum therapeutic range of tacrolimus trough concentrations in our transplant unit for the immunosuppressive regimen with MPA, tacrolimus, and steroid is 5-10 ng/mL for the 1st month after transplantation and 3-5 ng/mL thereafter, provided there is no evidence of rejection. Unlike other transplant patients, this patient was maintained on an immunosuppressive regimen of only tacrolimus and prednisolone, but at significantly higher tacrolimus trough concentrations than normal after stabilization (7-20 ng/mL). There were no clinical features of side effects including tremor or biochemical features of rejection or toxicity.
Mutations in the genes encoding for CYP3A5 and MDR 1 (2677G > T) explains the increased exposure to tacrolimus. The availability of this information in the early treatment period could have decreased the number of TDM specimens and if in future patients with this genotype can be managed on low doses of tacrolimus alone it will significantly reduce the cost of treatment. TDM is the proven cornerstone of dose individualization but pharmacogenomics has a vital role to play. In South Indians the frequency of CYP3A5*3 was found to be 63.5% which is significantly different from the incidence among the African Americans, Caucasians (87.3%), and Chinese population.[11]
Conclusion
This case report gives rise to a number of questions. Can other patients with this genotype be maintained on only low dose tacrolimus with steroids? Will a mandatory genetic screening of patients undergoing renal transplant reduce the overall cost and time to individualized therapy? Would it be necessary to define a different tacrolimus trough therapeutic range for patients maintained on only two drug immunosuppressive regimen (tacrolimus + steroid).
Acknowledgment
We would like to sincerely thank Dr. Poonkuzhali Balasubramanian and Savitha Varadarajan, Department of Hematology, Christian Medical College Hospital, Vellore for their help with pharmacogenomic analysis.
Source of Support: Nil
Conflict of Interest: None declared.
References
- Randomized trial of immunosuppressive regimens in renal transplantation. J Am Soc Nephrol. 2011;22:1758-68.
- [Google Scholar]
- Reduced exposure to calcineurin inhibitors in renal transplantation. N Engl J Med. 2007;357:2562-75.
- [Google Scholar]
- Explaining variability in tacrolimus pharmacokinetics to optimize early exposure in adult kidney transplant recipients. Ther Drug Monit. 2009;31:187-97.
- [Google Scholar]
- Hematocrit influences immunoassay performance for the measurement of tacrolimus in whole blood. Ther Drug Monit. 2005;27:766-9.
- [Google Scholar]
- Optimization of initial tacrolimus dose using pharmacogenetic testing. Clin Pharmacol Ther. 2010;87:721-6.
- [Google Scholar]
- CYP3A5 and ABCB1 polymorphisms and tacrolimus pharmacokinetics in renal transplant candidates: Guidelines from an experimental study. Am J Transplant. 2006;6:2706-13.
- [Google Scholar]
- Influence of ABCB1 polymorphisms and haplotypes on tacrolimus nephrotoxicity and dosage requirements in children with liver transplant. Br J Clin Pharmacol. 2009;68:413-21.
- [Google Scholar]
- AUC-guided dosing of tacrolimus prevents progressive systemic overexposure in renal transplant recipients. Kidney Int. 2005;67:2440-7.
- [Google Scholar]
- Therapeutic drug monitoring of mycophenolate mofetil in transplantation. Ther Drug Monit. 2006;28:145-54.
- [Google Scholar]
- A limited sampling strategy for tacrolimus in renal transplant patients. Br J Clin Pharmacol. 2008;66:467-72.
- [Google Scholar]
- Genetic polymorphisms of drug-metabolizing phase I enzymes CYP2E1, CYP2A6 and CYP3A5 in South Indian population. Fundam Clin Pharmacol. 2012;26:295-306.
- [Google Scholar]







