Translate this page into:
Cephalic Arch Stenosis: Location of Stenosis in Indian Hemodialysis Patients
This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
The cephalic vein is formed over the “anatomical snuff box” and joins the axillary vein just below the clavicular level. The definition of cephalic arch is varied. In the radiology literature, it is defined as the central perpendicular portion of the cephalic vein as it traverses the deltopectoral groove and joins the axillary vein. The possible etiologies of cephalic arch stenosis are numerous. This study aimed to identify patients with cephalic arch stenosis and to discern the domain site of stenosis. This is a retrospective case series of patients who had an arteriovenous fistula with dysfunction of access and ipsilateral upper-limb edema. The clinical features of the access dysfunction were strong pulse due to increased pressure, weak thrill due to poor proximal flow, high static pressure, or decreased dialysis efficiency. All these 25 patients underwent computed tomography (CT) angiogram. The CT angiographic findings revealed cephalic arch stenosis and stenosis in 13 patients (52%). domain IV was slightly more affected than other domains of cephalic arch.
Keywords
Cephalic arch stenosis
cephalic vein
computed tomography angiogram
ipsilateral upper-limb edema
vascular access dysfunction
Introduction
The cephalic vein, commonly formed over the “anatomical snuff box,” curves proximally from the radial end of the dorsal plexus round the forearm's radial side to its ventral aspect, receiving veins from both aspects. Distal to the elbow, a branch of the median cubital vein, joined by a ramus from the deep veins, diverges proximomedially to reach the basilic vein. The cephalic vein ascends in front of the elbow superficial to a groove between the brachioradialis and biceps, crosses superficially to the lateral cutaneous nerve of the forearm, ascends laterally to the biceps and between pectoralis major, pierces the clavipectoral fascia, crosses the axillary artery, and joins the axillary vein just below the clavicular level.[1] The definition of cephalic arch is varied. In the radiology literature, it is defined as the central perpendicular portion of the cephalic vein as it traverses the deltopectoral groove and joins the axillary vein.[2] Kian and Asif described it as the final arch of the cephalic vein before it joins the axillary vein.[3] In addition, “bifid” and “trifid” variants in which the arch forms two or three channels are also described.[4]
The possible etiologies of cephalic arch stenosis are pulsatile and increased pressure flow in fistulized cephalic vein, external compression by deltopectoral and claviculopectoral fascia and pectoralis major, the turbulence due to the angle of the arch which induces intimal hyperplasia, and the presence of valves in cephalic vein at least twice in number than in other veins.
In the forearm, radiocephalic arteriovenous fistulas (AVFs) drain through the basilic, brachial, and cephalic veins. On the Doppler ultrasound studies, only 30% of the total flow drains through the cephalic vein in the forearm, whereas the major portion of the flow drains through the basilic vein.[5] The more proximal brachiocephalic fistulas have a higher flow rate and in general, drain exclusively through the cephalic vein.
Bennett et al.[4] had standardized the definition of the cephalic arch and the location of the stenosis. Bennett et al.[4] had based their study on digital subtraction angiogram (DSA). The advantage of DSA is that a simultaneous endovascular intervention is possible. However, the perivenular pathologies are better evaluated on a computed tomography (CT) angiogram than on a DSA. The collaterals are definitely better visualized on DSA compared to CT angiogram, but CT angiogram with its multiplanar capability, reconstruction techniques, and latest postprocessing algorithms also demonstrates the collaterals adequately, which are clinically significant and necessary for decision-making. We identified the cephalic arch stenosis on a CT angiogram. DSA was not available at our institute. The standardization of location of stenosis would establish a reference for improved communication when cephalic arch stenosis is discussed.
This study aimed to identify patients with cephalic arch stenosis from our institute and to discern the site of stenosis domain according to the classification proposed by Bennett et al.[4]
Materials and Methods
Patients
This is a retrospective case series of patients who had an AVF with dysfunction of access and ipsilateral upper-limb edema. All patients were from a tertiary care institute in South India. The institute has 37 hemodialysis stations. The patients' data were maintained in a retrievable electronic and paper format. The clinical features of the access dysfunction, which raised red flags, included strong pulse due to increased pressure, weak thrill due to poor proximal flow, high static pressure, or decreased dialysis efficiency (calculated as Kt/V). All these patients underwent CT angiogram. Nonionic iodinated contrast, 100–120 mL of strength 300 mg/mL, was injected into the peripheral vein either in the contralateral upper limb if veins of only one limb are to be evaluated or into the peripheral veins of one of the lower limbs if veins in both upper limbs are to be evaluated. The rate of infusion was 3.5 mL/s. It was followed by a normal saline at a rate of 3.5 mL/s. The region of interest for determining the optimal opacification, i.e., 100 HU, was kept at the arch aorta (Boles tracking method). The images were acquired from the base of the skull to terminal phalanges. This was followed by venous-phase acquisition. The slice thickness was 5 mm and collimation was 0.6 mm. These images were reconstructed with a slice thickness of 2 mm. The postprocessing was done on “inspace” platform with maximum intensity projection and shaded surface display. Patients were excluded if there was anomalous venous anatomy such as a bifid arch (one), a trifid arch, or a chronic cephalic vein occlusion (two) that leads to several collaterals which replace cephalic arch. Presence of vessel wall thickening and narrowed caliber of the vessel and extensive collateralization favor chronicity of vessel obstruction, while the acute thrombosis results in distension of the vein.
Radiology
We followed the division of cephalic arch proposed by Bennett et al. The detailed description of division of cephalic arch is available elsewhere.[5] In brief, the cephalic arch was divided into four segments. First, a line was drawn through the arch apex perpendicular to the vein wall. Then, the distance from the cephalic-axillary vein junction to the apex was measured. By dividing the distance between the junction and the apex in half, we found the midpoint and inserted a line perpendicular to the arch at that location. The distance from midpoint to apex was used to place a third line perpendicular to the arch at a location equidistant beyond the apex. In the direction of normal blood flow through the cephalic vein, the four segments were labeled as I, II, III, and IV [Figure 1]. The diameter of the nearest normal cephalic vein peripheral to the arch was used as the standard by which to characterize the areas of narrowing to diagnose stenosis. In concordance with the DOQI 2006 AVF guidelines,[6] we considered significant stenosis to be 50% or more reduction in normal vein diameter. Two radiologists reviewed all the CT angiograms independent of each other and later discussed the differences in findings. They issued a common final report.

- Division of cephalic arch into four domains
Patients' data
Patients' age, sex, etiology of renal failure, history of diabetes, hypertension, coronary artery disease, peripheral vascular disease, duration of dialysis, duration after construction of AVF, and clinical features of access dysfunction were collected in a pro forma.
Results
From August 2013 to February 2016, there were 25 patients with end-stage renal disease on maintenance hemodialysis with vascular access dysfunction and ipsilateral upper-limb edema. The mean age of the patients was 51.4 years (range: from 17 to 72 years). There were 19 males. The primary kidney disease was secondary to diabetes mellitus in eight of them and the rest were nondiabetics. The mean dialysis vintage in the patient group was 26.64 months (range: from 3 to 68 months).
Of the 25 patients, 11 (44%) were having radiocephalic AVF (right upper limb 5, left upper limb 6), nine patients (36%) had brachiocephalic AVF (right upper limb 3, left upper limb 6), two patients (8%) had left brachiobasilic AVF, and three patients (12%) had brachioaxillary graft (right upper limb 2, left upper limb 1). Of the 25 patients, at the time of initiation of dialysis, 17 patients (68%) had noncuffed catheters and eight patients (32%) had native AVF. The site of first catheter insertion was the internal jugular vein (IJV) on the right side in a majority of them. One patient had a history of subclavian vein catheter insertion. In all patients, the first AVF was secured on the opposite side of IJV catheter placed. In no patient, the first AVF was secured on the upper limb as the same side of IJV catheter placed. This was in accordance with the National Kidney Foundation Kidney Disease Outcomes Quality Initiative (NKF KDOQI)™ guidelines.[6] Thirteen patients (52%) had a history of repeated catheter insertions at the same site. Nine patients (36%) had a history of multiple central vein catheter insertions at different sites. However, due to multiple vascular access failures, a second AVF was secured on the same side as that of previous IJV catheter placement. This practice should not have been implemented.
The mean catheter duration was 15.92 weeks. The NKF KDOQI™ guideline specifies that the noncuffed double-lumen internal jugular catheters should not be used for >3 weeks.[7] The presence of catheters for longer duration than this was fraught with the risks. At our institute, few patients required a catheter for exceptionally long durations. This was an undesirable practice. However, these patients had primary failures of multiple vascular access surgeries. Moreover, nine patients (36%) had a history of catheter-related bloodstream infection. It was likely related to prolonged presence of catheter.
All the 25 patients had ipsilateral limb edema. In addition, 9 (36%) patients had facial edema along with limb edema. The difficulty in cannulation was encountered in 13 (52%) patients. The venous pressures were raised during hemodialysis in 16 (64%) patients. There was difficulty in securing hemostasis at the end of dialysis procedure in 11 (44%) patients. The CT angiographic findings revealed cephalic arch stenosis in 13 patients (52%) of the 25 patients. The causes of edema of limb in the remaining patients were thrombosis of draining vein, steal syndrome, and pseudoaneurysms and aneurysms.
The mean age of these 13 patients was 50.3 years (range: 17–72 years). There were nine males and five diabetics. The main presenting features of these 13 patients were ipsilateral limb edema (13), difficulty in cannulation (8), ipsilateral facial edema (2), raised venous pressures during hemodialysis (8), difficulty in securing hemostasis at the end of dialysis (5), and a combination of all these features (6). Of the 13 patients, seven (53.8%) were having radiocephalic AVF (right upper limb 2, left upper limb 5) and six patients (46.2%) had brachiocephalic AVF (right upper limb 2, left upper limb 4).
The mean duration for development of symptoms secondary to cephalic arch stenosis was 23.15 months (range: 9–48 months). It did not differ between diabetic and nondiabetic patients. Table 1 and Figures 2–5 show the site of stenosis in these 13 patients.


- Stenosis in segments II, III, and IV
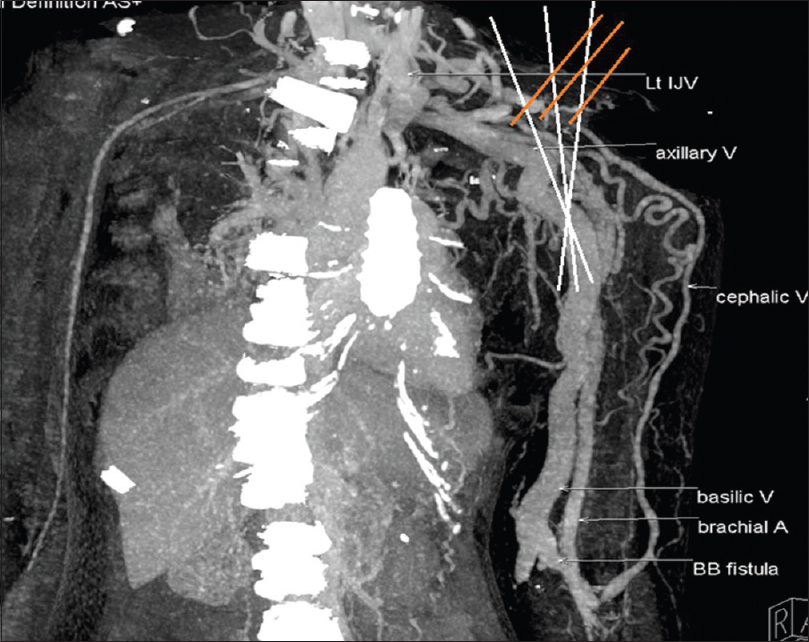
- Stenosis in segments II, III, and IV

- Stenosis in segment III

- Stenosis in segments III and IV
Discussion
The published articles on cephalic arch stenosis are sparse. This article is the first one that presented data on cephalic arch stenosis in Indian hemodialysis patients. We followed the division of cephalic arch as proposed by Bennett et al.[4] In our patients, there was not much difference in domains of the cephalic arch affected; domain IV was slightly more affected than other domains of cephalic arch. In the study of Bennett et al.,[4] domain IV was the most common domain to have evidence of stenosis (72.5%) followed by domain III (56%), domain II (40.6%), and then domain I (17.4%).
Bennett et al.[4] identified cephalic arch stenosis with the help of DSA. We used CT angiogram with comparable results. The data of domain involvement of cephalic arch by either of these two methods when accumulated from several institutes would have benefits. The identification of the site of stenosis in various domains guides us toward the role of valves. There were 2–9 valves in the cephalic vein, the highest when compared to the basilic (1–6) and the brachial veins (1–5).[7] The valves in the cephalic vein are distributed along the vein, but they are most frequent at the area between the upper margin and lower margin of the cubital fossa, the part from the distal 1/3rd of the forearm to the styloid processes of the radius and the ulna. In addition, there is a valve at the insertion of the cephalic vein to axillary vein in 8% of individuals.[8]
The studies have identified more frequent cephalic arch stenosis in patients with brachiocephalic AVF than radiocephalic AVF, and diabetic patients appeared less disposed to cephalic arch stenosis.[910] A patient with a dysfunctional brachiocephalic fistula is 37 times more likely to harbor cephalic arch stenosis than a patient with a dysfunctional radiocephalic fistula.[2] In the study of Hammes et al., cephalic arch stenosis was present in 33 nondiabetics and 12 diabetics. In our study, there were no differences in patients with cephalic arch stenosis among these parameters. It might be related to a small number of patients.
The options available for management were angioplasty, stent placement, cutting balloon angioplasty, inflow reduction, and surgeries such as cephalic vein turn down and patch angioplasty.[11] There is a possibility that optimal treatment may vary by the domain affected. The domain affected may have an effect on the rates of resistance to angioplasty, inflation pressures required, rupture rates during angioplasty, and use of stent with angioplasty. A trial comparing these options for each domain is needed. For such a trial, a better communication based on a classification is needed.
This study had a few limitations. They include retrospective design and small number of patients.
Conclusion
We must highlight that it is prudent to assess for central venous stenosis before securing an AVF on the ipsilateral side of a central vein catheter placement.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Gray's Anatomy (37th ed). UK, Edinburgh: Churchill Livingstone; 1989. p. :806.
- Prevalence and treatment of cephalic arch stenosis in dysfunctional autogenous hemodialysis fistulas. J Vasc Interv Radiol. 2003;14:567-73.
- [Google Scholar]
- Characterization of the cephalic arch and location of stenosis. J Vasc Access. 2015;16:13-8.
- [Google Scholar]
- Managing Cephalic Arch Stenosis. Endovascular Today; 2012. p. :63-5.
- Clinical practice guidelines for vascular access. Am J Kidney Dis. 2006;48(Suppl 1):S248-73.
- [Google Scholar]
- NKF-K/DOQI clinical practice guidelines for vascular access: Update 2000. Am J Kidney Dis. 2001;37:S137-81.
- [Google Scholar]
- Anatomy of arm veins: Significance for vein valve transplantation. Clin Anat. 1992;5:45-9.
- [Google Scholar]
- Cephalic arch stenosis in dialysis patients: Review of clinical relevance, anatomy, current theories on etiology and management. J Vasc Access. 2014;15:157-62.
- [Google Scholar]
- Characteristic differences in cephalic arch geometry for diabetic and non-diabetic ESRD patients. Nephrol Dial Transplant. 2009;24:2190-4.
- [Google Scholar]
- Cephalic arch stenosis: Mechanisms and management strategies. Semin Nephrol. 2012;32:538-44.
- [Google Scholar]







