Translate this page into:
Maternal drugs and neonatal renal failure
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Maternal use of drugs during pregnancy may cause irreversible renal failure in the newborn. This report highlights the adverse effect of telmisartan during the last trimester of pregnancy. The neonate presented with oliguric renal failure and the renal histology showed proximal tubular dysgenesis.
Keywords
Neonatal renal failure
pregnancy
proximal tubular dysgenesis
renin angiotensin system blockade
Introduction
Inappropriate drug use during pregnancy may lead to fetal complications. Use of angiotensin receptor blockers (ARBs) in the third trimester may cause renal failure in the neonate which may be irreversible if associated with renal tubular dysgenesis. This case report highlights the importance of educating the practitioners about appropriate use of drugs in pregnancy.
Case Report
A 7-day-old male child was brought to our hospital with severe respiratory distress and anuria. Child was born to non-consanguineous parents and was 2nd in birth order. The older female sibling was normal. The mother had pregnancy induced hypertension diagnosed at 21st week of gestation and review of her pregnancy medications showed that she had been taking telmisartan for hypertension as prescribed by a local practitioner. Her blood pressure was well controlled. Proteinuria was 550 mg/d and serum creatinine was 0.8 mg/dl. Her antenatal scan showed oligohydramnios, no fetal anomalies were detected. She underwent caesarean section at 34 weeks. The birth weight of the newborn was 2.5 kg. The child had a progressive decline in urine output and was brought to the hospital for respiratory distress on 7th day of life.
Examination showed no congenital abnormalities. The child has severe respiratory distress. Urine examination was normal, serum creatinine was 8 mg/dl, blood urea 148 mg/dl, serum potassium 6 meq/l, serum sodium 135 meq/l, serum bicarbonate 12 meq/l, blood sugar 120 mg/dl, hemoglobin 13 g/dl, total leucocyte count was 7800/mm3 with normal differential count and C-reactive protein was negative. Urine and blood cultures were sterile. Chest radiography and echocardiogram were normal. On ultrasound examination right kidney measured 3.8 cm × 2 cm and left kidney was 4 cm × 1.8 cm with grade 1 echotexture. The neonate was subjected to peritoneal dialysis. The parameters settled. However the child continued to be anuric. Another peritoneal dialysis was carried out. In view of non-recovery of renal functions a renal biopsy was performed at 21st day of life which revealed proximal tubular dysgenesis (PTD) characterized by glomerular overcrowding on hematoxylin and eosin stain with immunostaining demonstrating absence of proximal tubular antigens (CD 10 negative). There was normal staining for distal tubular antigens (epithelial membrane antigen) [Figures 1-3]. The child was initiated on continuous ambulatory peritoneal dialysis. This case highlights an important and preventable cause of neonatal renal failure.
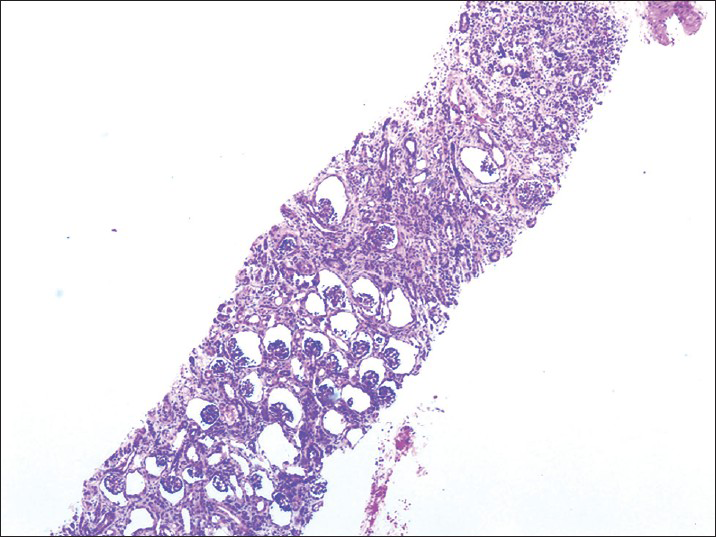
- Proximal tubular dysgenesis with glomerular crowding (H and E, ×10)

- Immunostaining-absence of proximal tubules (CD 10 negative, ×40)

- Immunstaining-normal distal tubules (stain for epithelial membrane antigen positive, ×40)
Discussion
The first detailed description of this disorder was given by Schwartz et al.[1] It was later described by Voland et al.,[2] Allanson et al.[3] and Swinford et al.[4] The patient reported by Lorentz and Trillo with glomerular as well as tubular immaturity represents a different disorder.[5]
PTD may occur due to congenital or acquired causes. Congenital defects result from mutations in genes of the renin angiotensin system (RAS) that encode genes for renin, angiotensinogen (AGT), angiotensin-converting enzyme (ACE) and Ang II receptor type 1 and are associated with autosomal recessive PTD.[23] These mutations result in absent or ineffective Ang II, leading to a loss of negative regulation of renin synthesis. Low Ang II action causes persistent hypotension during fetal development. Chronic under perfusion pressure in the kidney causes a defect in the proximal tubule development.
Acquired causes of PTD include neonatal hemochromatosis (NH), substance abuse and RAS blockade and twin-twin transfusion syndrome (TTTS).
The fetal liver injury in NH impairs proximal renal tubular development through impaired hepatic AGT production. A linear relationship is seen between hepatic AGT expression and degree of renal tubular dysgenesis. Vascular impairment due to cocaine or alcohol intake in mother or maternal smoking may lead to PTD. Angiotensin-converting enzyme inhibitor (ACEi) intake by the mother is an important causative factor.[6] RAS system is critical for renal development. Maternal use of ACEi or ARBs leads to reduction in angiotensin or its’ action. Use of these drugs in the first trimester is associated with cardiovascular or central nervous system anomalies in 5%. Use in 2nd or 3rd trimester is absolutely contraindicated as it leads to fetal anuria, oligohydramnios, pulmonary hypoplasia, Potter's phenotype, hypocalvaria, refractory hypotension and even death. TTTS in monochorionic twins may be associated with PTD. There is a disparity in circulation with discordant fetal growth, urine output and amniotic fluid accumulation and hypoperfusion due to shunting of blood from donor to recipient twin leading to PTD in the donor twin.
Some mothers have spontaneous abortions if the fetus is affected with PTD. PTD is characterized by oligohydramnios in mother.[46] However, normal amniotic fluid volume is seen even in affected pregnancies prior to the 20th or 22nd week of gestation hence early prenatal diagnosis is difficult. Late second trimester demonstration of oligohydramnios on ultrasound with normal sized fetal kidneys suggests this diagnosis. Severe oligohydramnios may be seen. The children may have Potter's phenotype (redundant skin, facial dysmorphism with large and flattened low-set ears, arthrogryposis and severe deformation of the head, chest, hips and limbs) and anuric renal failure leading to stillbirth or neonatal death from respiratory failure. There may be microcephaly, congenital heart defect, acidosis, joint contractures or fractures. Many are born preterm. Fetal chromosomes appear normal. Severe and refractory hypotension has been observed in children who survive the immediate neonatal period. Poor calvarial ossification, resulting in wide cranial sutures and large fontanels, is an additional feature.
The gross appearance of the kidneys is unremarkable. The cortex maybe slightly thinned and the medulla may be a little attenuated. The kidneys show normal fetal lobulation. There are no gross abnormalities in the cardiovascular, gastrointestinal, endocrine, reticuloendothelial, or central nervous systems. The renal cortices may be slightly pale, but the kidneys and the rest of the genitourinary system are otherwise grossly normal. Kidneys are normal in size, shape and echogenicity or may show mild hyperechogenicity and/or poor corticomedullary differentiation with enlargement.
Histologically patients demonstrate a paucity of proximal tubules with crowding of glomeruli. There is a relative and an absolute increase in numbers of normal glomeruli. Kidney shows a lack of ‘mature proximal tubules on immunostaining with fumarylacetoacetate hydrolase, Leu-M1 and Lotus tetragonolobus which are used to identify proximal convoluted tubules. Monoclonal anti-CD10 antibody that recognizes the human membrane-associated neutral endopeptidase of podocytes and proximal tubular cells and a monoclonal anti-CD15 antibody that recognizes an early myeloid differentiation antigen and serves as a marker for proximal tubules show paucity of proximal tubules. There is a positive staining of distal convoluted and collecting tubules with epithelial membrane antigen. The kidneys show tubular immaturity with some increase in interstitial fibrous tissue. The immature tubules have hyperchromatic nuclei, prominent nucleoli; scant cytoplasm and their brush borders are negative to periodic acid-Schiff reagent. There is no evidence of renal dysplasia. In some cases, there is an arrest of glomerular maturation. The cortical and medullary capillary network can be identified by staining with anti-CD34 antibodies and is normal in PTD. Renal vein thrombosis has been reported in few cases.[7] ACE and AGT are absent or only very weakly expressed in kidney tubules.[8]
Acknowledgments
The authors would like to thank Dr. Swarnalata Gowrishankar, Dr. Michelle de Padua and Dr. Meenakshi Swain, Pathology Department, Apollo Hospital, Hyderabad for the histopathology slides.
Source of Support: Nil
Conflict of Interest: None declared.
References
- Isolated congenital renal tubular immaturity in siblings. Hum Pathol. 1986;17:1259-63.
- [Google Scholar]
- Congenital hypernephronic nephromegaly with tubular dysgenesis: A distinctive inherited renal anomaly. Pediatr Pathol. 1985;4:231-45.
- [Google Scholar]
- Possible new autosomal recessive syndrome with unusual renal histopathological changes. Am J Med Genet. 1983;16:57-60.
- [Google Scholar]
- Renal tubular dysgenesis: Delayed onset of oligohydramnios. Am J Med Genet. 1989;32:127-32.
- [Google Scholar]
- Renal tubular dysgenesis (RTD)-an important cause of the oligohydramnion-sequence. Report of 3 cases and review of the literature. Pathol Res Pract. 2000;196:861-5.
- [Google Scholar]
- Renal tubular dysgenesis with fetal renal vein thrombosis. Pathologe. 1995;16:143-7.
- [Google Scholar]
- Renal tubular dysgenesis, a not uncommon autosomal recessive disorder leading to oligohydramnios: Role of the renin-angiotensin system. J Am Soc Nephrol. 2006;17:2253-63.
- [Google Scholar]







