Translate this page into:
Spectrum of Hypokalemic Paralysis from a Tertiary Care Center in India
This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Hypokalemic paralysis is an important and reversible cause of acute flaccid paralysis. The treating physician faces unique diagnostic and therapeutic challenges. We did a prospective study and included all patients with acute flaccid weakness and documented serum potassium of <3.5 mEq/L during the period between January 2009 and August 2015. We studied the incidence, etiology, clinical profile, and acid–base disturbances in patients presenting with hypokalemic paralysis and analyzed the significance of periodic and non-periodic forms of hypokalemic paralysis on patient's outcome. Two hundred and six patients were studied with a mean follow-up of 3.6 ± 1.2 years. Mean age was 37.61 ± 2.2 years (range 18–50 years). Males were predominant (M:F ratio 2.1:1). The nonperiodic form of hypokalemic paralysis was the most common (61%). Eighty-one (39%) patients had metabolic acidosis, 78 (38%) had normal acid–base status, and 47 (23%) patients had metabolic alkalosis. The most common secondary cause was distal renal tubular acidosis (RTA) (n = 75, 36%), followed by Gitelman syndrome (n = 39, 18%), thyrotoxic paralysis (n = 8, 4%), hyperaldosteronism (n = 7, 3%), and proximal RTA (n = 6, 4%). Patients with non-periodic paralysis had more urinary loss (40.1 vs. 12.2 mmol, P = 0.04), more requirement of potassium replacement (120 vs. 48 mmol, P = 0.05), and longer recovery time of weakness (48.1 vs. 16.5 h, P = 0.05) than patients with periodic paralysis. Non-periodic form of hypokalemic paralysis was the most common variant in our study. Patients with periodic paralysis had significant incidence of rebound hyperkalemia.
Keywords
Acute flaccid paralysis
distal renal tubular acidosis
Gitelman syndrome
hypokalemia
Introduction
Hypokalemic paralysis represents a group of heterogeneous disorders that present with acute flaccid muscle weakness and hypokalemia. If untreated, complications secondary to hypokalemia such as a cardiac arrhythmia or respiratory paralysis lead to morbidity and mortality.[12] In Asian population, the most common cause is thyrotoxic hypokalemic paralysis, whereas familial periodic paralysis is the leading cause in Caucasians.[34] Hypokalemia and paralysis can be divided into two types, hypokalemic periodic paralysis where there is transcellular shift of potassium into cells and hypokalemic non- periodic paralysis resulting from a large deficit of potassium due to various etiologies.
Materials and Methods
We conducted this prospective study in the Institute of Nephrology, Rajiv Gandhi Government General Hospital, Chennai, from January 2009 to August 2015. Patients admitted with acute onset of flaccid weakness and documented serum potassium of <3.5 mEq/L during the episode were included in the study after getting informed consent. Patients with renal dysfunction (serum creatinine >1.5 mg/dl) and pregnancy were excluded from the study. The institutional ethics committee approval was obtained.
The clinical data included age, sex, and history regarding the evolution of the symptoms, precipitating factors such as strenuous exercise, heavy carbohydrate meal in the preceding 24 h, alcohol consumption, and treatment details. Any similar illness in the family was enquired and reports of weakness, thyroid disease, diarrhea, vomiting, hypertension, bone pain, fractures, dry mouth, dry eyes, and renal stone disease were also recorded. Intake of drugs such as diuretics, β2-agonists, decongestants, insulin, laxatives, and antipsychotics was noted. The clinical examination included anthropometry, pulse, blood pressure, and all systems examination. Schirmer's test was performed in selected patients. Arterial blood gas analysis was performed by ABL800 (Radiometer, Denmark). Urine and plasma biochemical studies were conducted by automated analyzer (Microlab300 Merck, Germany).
Case definitions
Hypokalemic periodic paralysis[3]
Patients who had normal acid–base balance and without renal loss of potassium (spot urine potassium <15 mmol/L) were considered to have hypokalemic periodic paralysis.
Hypokalemic non-periodic paralysis[3]
Patients who had acid–base disturbance (metabolic acidosis or metabolic alkalosis) with renal loss of potassium were grouped under hypokalemic non-periodic paralysis.
Renal tubular acidosis[5]
RTA was diagnosed when patients with hyperchloremic metabolic acidosis with normal anion gap in the absence of gastrointestinal loss of potassium were found to have a urine pH more than 5.5. The diagnosis of proximal RTA (pRTA) is supported by the presence of glycosuria, aminoaciduria, and phosphaturia. The diagnosis of dRTA is supplemented by the presence of nephrocalcinosis in an X-ray kidney, ureter, and bladder/ultrasonogram. Sjogren's syndrome was diagnosed with positive lip biopsy, Schirmer's test, and serology test.
Gitelman syndrome[6]
Patients with normotension, metabolic alkalosis, and hypomagnesemia with hypocalciuria (urine calcium–creatinine ratio <0.2) were considered to have Gitelman syndrome.
Sporadic periodic paralysis
Those with normal acid–base balance with no documented renal loss of potassium in the presence of normal thyroid function.
Thyrotoxic periodic paralysis
Those with normal acid–base balance with no documented renal loss of potassium in the presence of thyrotoxicosis.
Hyperaldosteronism
Patients with hypertension, hypokalemia, and metabolic alkalosis with high aldosterone and low renin levels.
Liddle's syndrome
Patients with hypertension, hypokalemia, and metabolic alkalosis with low aldosterone and renin levels.
Statistical analysis
All data are expressed as mean ± standard deviation differences in group means between two variables were compared using t-tests. Differences in categorical variables were compared using Fisher's exact test. The difference was considered statistically significant if P < 0.05. Data were analyzed using SPSS Statistics for Windows Version 17.0 Chicago: SPSS Inc.
Results
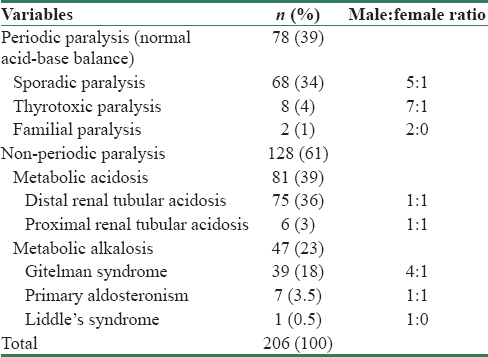
Two hundred and six patients with a mean age of 37.61 ± 1.2 years (range 18–50 years) were included in the study. Male: female ratio was 2.1:1 (140:66). Patients were grouped as periodic and non-periodic paralysis based on acid–base status as shown in Table 1.
Hypokalemic periodic paralysis
Seventy-eight patients with normal acid–base balance and no evidence of renal potassium loss (urine spot potassium <15 mmol/L) were considered to have hypokalemic periodic paralysis. Eight (4%) among them had biochemical evidence of thyrotoxicosis and were diagnosed as thyrotoxic periodic paralysis. Two patients with positive family history (father–son duo) were grouped under familial periodic paralysis and 68 (34%) patients who had a normal thyroid profile and negative family history of periodic paralysis were diagnosed as sporadic periodic paralysis. The mean potassium at presentation, rate of recurrence, incidence of respiratory paralysis, mean potassium requirement, and recovery time are tabulated in Table 2.

Hypokalemic non-periodic paralysis
Patients in this cohort had acid–base disorder, and they were classified into two groups, namely, metabolic acidosis and metabolic alkalosis. Among the 81 patients with metabolic acidosis, all had evidence of renal potassium loss (spot urine potassium >15 mmol/L). Six patients were diagnosed to have pRTA based on features of generalized proximal tubular dysfunction and urine pH was <5.5 during acidemia. Seventy-five patients (36%) had urine pH >5.5 during acidemia and were diagnosed to have dRTA. Forty-seven patients (23%) had metabolic alkalosis with renal potassium wasting. Diuretic use was excluded by careful scrutiny. Among them, 39 patients (16%) were normotensive and had hypocalciuria and hypomagnesemia. They were diagnosed to have Gitelman syndrome. Seven patients (4%) who had hypertension with high aldosterone with low renin levels were clubbed under hyperaldosteronism. Two among them had adrenal adenoma. One patient who had hypertension with low plasma renin activity and aldosterone was diagnosed as Liddle's syndrome.
All patients with hypokalemic paralysis were treated with intravenous or oral potassium chloride therapy ranging from 40 to 160 mEq/L. The mean potassium each patient received till recovery was 84 mEq/L. There was no significant difference in serum potassium level at the time of presentation between the periodic and non-periodic groups as shown in Table 3. However, there was a significant difference in urinary potassium loss, potassium requirement, and recovery time between the periodic and non-periodic forms of paralysis. There was a significant incidence of rebound hyperkalemia in patients with periodic paralysis. Patients of Gitelman syndrome with concurrent hypomagnesemia also received magnesium supplements.

Discussion
Hypokalemia and paralysis can be divided into two types, hypokalemic periodic paralysis where there is redistribution of potassium into cells and hypokalemic non-periodic paralysis resulting from a large deficit of potassium due to various etiologies. The differential diagnosis in a patient with hypokalemic paralysis can be challenging due to the heterogeneity of its etiologies, but it is important to make the diagnosis promptly because different therapies are required for each type and identifying causes that are reversible is important. The presence of a positive family history and recurrent episodes in a patient can be helpful in making a diagnosis of periodic paralysis, but periodic and non-periodic forms cannot be distinguished on clinical features alone.[78]
RTA was the most common cause of hypokalemic paralysis in our series, contributing to 40% of all patients. Table 4 compares this study with the available literature worldwide.[9101112] dRTA as a cause of hypokalemic paralysis in our study is similar to various published Indian studies (40%–69%).[14] dRTA accounts for 93% and pRTA was seen in 7%. The most common cause of dRTA in our series was Sjogren's syndrome. Ram et al. in their series of 152 consecutive cases of RTA, have documented Sjogren's syndrome as a major cause (34.8%).[15] Of these, 30 patients had dRTA, while 22 had pRTA. Around 23% of their cohort presented with hypokalemic paralysis. This study validates the observation that hypokalemic paralysis in Sjogren's syndrome may precede classical clinical description and can serve as the clinical marker for diagnosis.[1617]

We noted a high incidence of Gitelman syndrome (19%) in our series. The published studies had reported an incidence of 1%–9%.[61718] Males were predominantly affected (male:female ratio 4:1). As Gitelman syndrome is characterized by high phenotypic variability and clinical manifestations may vary from nonspecific symptoms to severe disability, there has been significant underdiagnosis of Gitelman syndrome.[619] We diagnosed more patients with Gitelman syndrome as the study was carried out in the nephrology department when compared to other studies from India where it was done in neurology department. Genetic studies and long-term follow-up will be required to map the higher prevalence of Gitelman syndrome in our population. Among the various etiologies of hypokalemic paralysis in our series, Gitelman syndrome has less recurrence, implying good response to treatment.
Thyrotoxic periodic paralysis was the most common cause of hypokalemic paralysis reported in various studies in the non-Caucasian population, accounting for nearly 50% of all patients. We noted only eight patients (4%) of thyrotoxic periodic paralysis in our series which was consistent with other Indian studies.[91012] The similarities in the present study and previous studies were male preponderance, lesser potassium requirement, and quick recovery.
In the present study, we had seven patients (3.5%) with features of hyperaldosteronism, which was consistent with other studies, with a reported incidence between 2% and 6%. Two had adrenal adenoma and underwent adrenalectomy, while the rest were treated with spironolactone in maximum tolerated doses. This cohort required more potassium replacement and needed more time for recovery when compared to the other cohorts. Only one study from India has reported a higher proportion of hyperaldosteronism (42%) as a cause of hypokalemic paralysis. However, that series involved only 31 patients from a tertiary endocrine center.[9]
About 34% patients who had normal acid balance with no renal cause of potassium loss with normal thyroid function have been clustered under sporadic hypokalemic paralysis. This has been consistent with other studies, which reported incidence between 10% and 57%. This cohort required less potassium replacement, less recovery time, and more chances of rebound hyperkalemia. Hence, recognition of this cohort is important to prevent fatal hyperkalemia.
Patients with non-periodic paralysis had 4-fold higher urinary potassium loss (40 vs. 12.2 mmol/L, P = 0.04) and required three times more potassium replacement (120 vs. 48 mmol/L, P = 0.05) and took 3-fold longer hours (48 vs. 16.5 h, P = 0.05) to recover than patients with periodic paralysis. This observation concurs with similar findings by Sung et al.[11] Overzealous replacement of potassium results in rebound hyperkalemia in patients with periodic paralysis, whereas none of the patients with non-periodic paralysis had rebound hyperkalemia (12 vs. 0, P = 0.0001). Sung et al.[11] reported paradoxical hypokalemia in patients with non-periodic paralysis, which refers to paradoxical fall in serum potassium while on potassium replacement for severe hypokalemia. Although paradoxical hypokalemia is reported in both periodic and non-periodic paralysis, it is more common with non-periodic paralysis. The reason attributed was uncorrected volume depletion causing sustained renin-aldosterone activation causing hypokalemia.
Our study highlights the need to identify secondary causes of hypokalemic paralysis as they need specific therapies besides the high prevalence of RTA and Gitelman syndrome in the Indian population. Prompt recognition and management will prevent incapacitating quadriparesis and loss of quality of life. Failure to address the underlying cause of hypokalemia will lead to aggressive therapy, putting patients at risk for fatal hyperkalemia.[20]
Limitations and strengths
We have not measured 24 h urine potassium excretion in our study; however, Lin et al. have shown measuring urine spot potassium to be equally effective.[13] Secondly, genetic analysis could not be done in any of the subjects due to logistic reasons, and thirdly being a tertiary referral center, in most of our patients, hypokalemia was severe and/or recurrent. The strengths of this study are its prospective nature with the largest number of individuals ever included, besides the long follow-up period.
Conclusions
Non-periodic paralysis was the most common form of hypokalemic paralysis in our study. dRTA was the most common secondary cause of hypokalemic paralysis followed by Gitelman syndrome. Patients with non-periodic paralysis had more urinary potassium loss, a higher potassium requirement, and longer recovery time. Patients with periodic paralysis had significantly higher incidence of rebound hyperkalemia.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgment
This study was supported by academic research grant from The Tamil Nadu Dr MGR Medical University, Chennai (TNMMU-project number 43/2011).
References
- Disorders of potassium homeostasis: An approach based on pathophysiology. Am J Kidney Dis. 1994;24:597-613.
- [Google Scholar]
- Hypokalemic paralyses: A review of the etiologies, pathophysiology, presentation and therapy. Am J Emerg Med. 1992;10:143-8.
- [Google Scholar]
- Pathophysiology and management of hypokalemia: A clinical perspective. Nat Rev Nephrol. 2011;7:75-84.
- [Google Scholar]
- Clinical and pathophysiologic spectrum of acquired distal renal tubular acidosis. Kidney Int. 1981;20:389-96.
- [Google Scholar]
- Gitelman syndrome: Consensus and guidance from a kidney disease: Improving global outcomes (KDIGO) controversies conference. Kidney Int. 2017;91:24-33.
- [Google Scholar]
- Aetiological, clinical and metabolic profile of hypokalaemic periodic paralysis in adults: A single-centre experience. Natl Med J India. 2006;19:246-9.
- [Google Scholar]
- Spectrum of hypokalaemic periodic paralysis in a tertiary care centre in India. Postgrad Med J. 2010;86:692-5.
- [Google Scholar]
- Etiologic and therapeutic analysis in patients with hypokalemic nonperiodic paralysis. Am J Med. 2015;128:289-960.
- [Google Scholar]
- Hypokalaemic periodic paralysis in rural northern India–Most have secondary causes. Trop Doct. 2014;44:33-5.
- [Google Scholar]
- Laboratory tests to determine the cause of hypokalemia and paralysis. Arch Intern Med. 2004;164:1561-6.
- [Google Scholar]
- Clinical and biochemical spectrum of hypokalemic paralysis in north: East India. Ann Indian Acad Neurol. 2013;16:211-7.
- [Google Scholar]
- Renal tubular acidosis in Sjögren's syndrome: A case series. Am J Nephrol. 2014;40:123-30.
- [Google Scholar]
- Primary sjogren's syndrome presenting as hypokalemic periodic paralysis and respiratory arrest. Clin Rheumatol. 2001;20:365-8.
- [Google Scholar]
- Hypokalemic periodic paralysis in Sjögren's syndrome. Arch Intern Med. 1981;141:1671-3.
- [Google Scholar]
- A new familial disorder characterized by hypokalemia and hypomagnesemia. Trans Assoc Am Physicians. 1966;79:221-35.
- [Google Scholar]
- Hypokalaemia and subsequent hyperkalaemia in hospitalized patients. Nephrol Dial Transplant. 2007;22:3471-7.
- [Google Scholar]







