Translate this page into:
Adenovirus Interstitial Nephritis: An Unusual Cause for Early Graft Dysfunction
This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
We describe a rare case of adenovirus interstitial nephritis in a 37-year-old man, 4 weeks following deceased donor renal transplantation. He presented with gross hematuria and acute graft dysfunction. A renal biopsy revealed necrotizing tubulointerstitial nephritis with intranuclear viral inclusions in the tubular epithelial cells. Immunohistochemistry and polymerase chain reaction confirmed adenovirus infection. Reduction in immunosuppression alone resulted in rapid improvement of graft function. Awareness of the clinical and characteristic biopsy findings may help establish the correct diagnosis, which is crucial as disseminated infection, if left untreated, is associated with a high mortality rate in renal allograft recipients.
Keywords
Acute graft dysfunction
adenovirus
interstitial nephritis
renal biopsy
Introduction
Viral infections can cause significant morbidity in renal transplant recipients. BK polyomavirus (BKV) and cytomegalovirus (CMV) are the more frequently encountered viral infections. Adenovirus interstitial nephritis (ADVIN) of the renal allograft is an exceptionally rare occurrence. We describe a case of ADVIN that presented within four weeks after deceased donor renal transplantation.
Case Report
A 37-year-old male with end-stage renal disease due to chronic glomerulonephritis on maintenance hemodialysis underwent deceased donor renal transplantation in March 2015. Complement-dependent cytotoxicity crossmatch was negative and CMV serology status was donor negative/recipient positive. Basiliximab (interleukin-2 receptor antagonist 20 mg 2 doses on day 0 and day 4) was used for induction along with methylprednisolone 1 g for 3 days, followed by triple immunosuppression (tacrolimus 0.1 mg/kg/day, mycophenolate mofetil [MMF] 2 g/day, and prednisolone 20 mg/day).
He underwent reexploration on the same day for a bleeding complication. There was no vascular anastomotic leak. He was transfused with three units of leukocyte filtered packed red blood cells. During the course, he developed lower respiratory tract infection (sputum culture - Klebsiella), which was treated with intravenous (IV) meropenem and supportive treatment.
He reached a best creatinine of 1.0 mg/dl by 10th postoperative day. As his tacrolimus trough level on the 10th day was 3.76 ng/ml with a dose of 0.1 mg/kg/day in two divided doses, tablet diltiazem 30 mg once daily was added, and the tacrolimus trough level reached 7.7 ng/ml on the 15th day. He was discharged with the triple drug regimen, a beta blocker, antiviral, and antibiotic prophylaxis (Valganciclovir 450mg and Cotrimoxazole double-strength once daily).
On day 23 posttransplantation, he started passing blood clots in the urine. There was no dysuria or fever and his creatinine was 1.5 mg/dl. The double J-stent in situ was removed on day 25. The next day, his creatinine increased to 1.8 mg/dl and the hematuria persisted. Urine culture following stent removal grew Klebsiella. He was given IV antibiotics for the positive urine culture though the white blood cell count was normal. Procalcitonin level was 0.24 ng/ml. Graft ultrasound revealed normal echotexture, no hydroureteronephrosis, and a resistive index of 0.6 units. The possibility of a viral infection was considered. Urine for Decoy cells was negative. Adenovirus DNA was detected in the urine by polymerase chain reaction (PCR) using in vitro diagnostic approved Multiplex PCR Kit targeting human adenovirus using specific primers/probe, Fast Track Diagnostics, Luxembourg. PCR was carried out in Rotor-Gene Q 5Plex real-time PCR machine, Qiagen, Germany; PCR for CMV and BKV was negative.
The renal allograft biopsy showed edematous cortex and medulla diffusely infiltrated by lymphocytes, plasma cells, and neutrophils. Areas of interstitial hemorrhage were present. There was tubular epithelial cell necrosis and cell debris filled the tubular lumen [Figure 1]. Some of the tubular basement membranes were ruptured. Foci of tubular epithelial cells had enlarged nuclei with smudged nuclear material [Figure 2]. No granulomas were identified. The glomeruli and blood vessels were spared. Immunostaining for C4d was negative. Immunohistochemical staining for adenovirus (anti-adenovirus antibody clone 20/11, dilution 1:400, EMD Millipore, Germany) was positive on the abnormal tubular epithelial cells [Figure 3]. Immunohistochemical stains for CMV and BKV were negative.
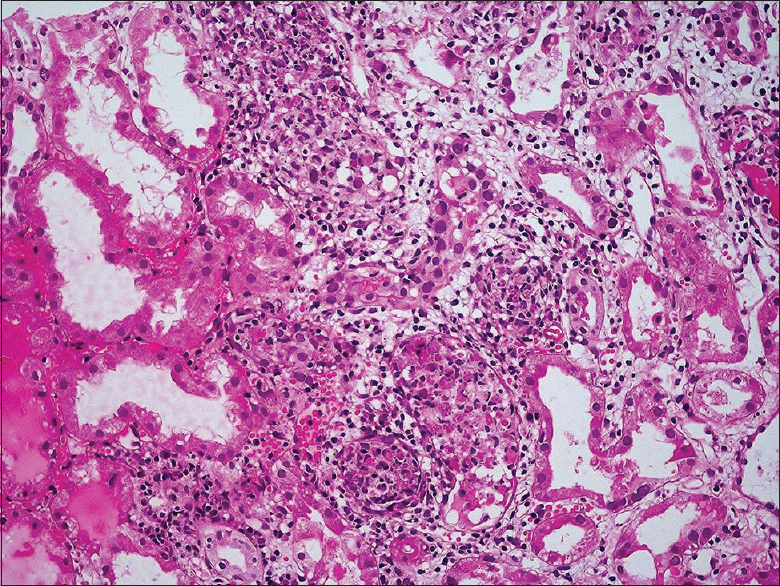
- Focal necrotizing tubulointerstitial nephritis (H and E, ×200)
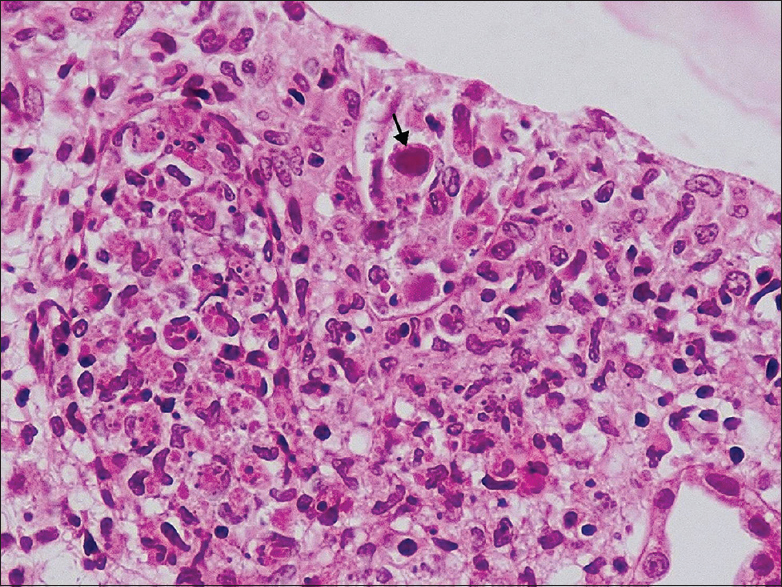
- Intranuclear viral inclusion body (arrow) in the tubular epithelial cell (H and E, ×400)
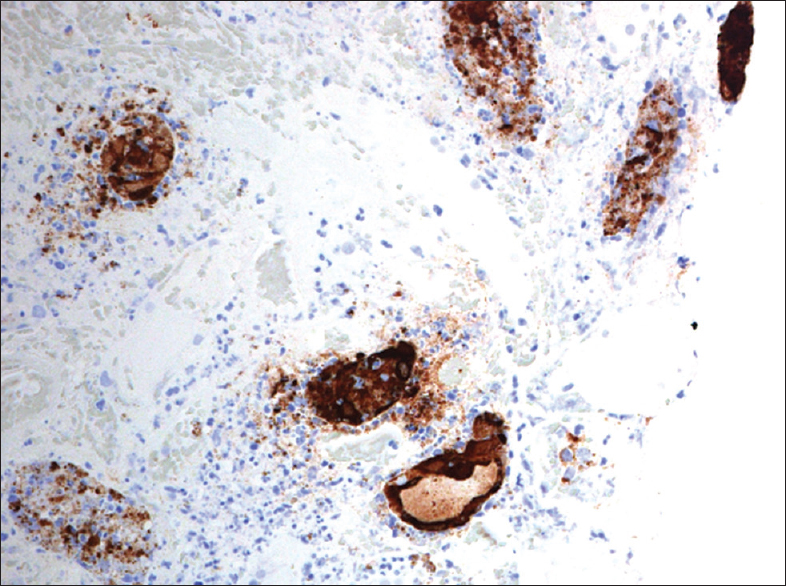
- Immunohistochemical stain for adenovirus antigen shows intranuclear and cytoplasmic staining in the tubular epithelial cells
The diagnosis of ADVIN was made based on viral cytopathic effects seen on light microscopy, presence of viral antigens by immunohistochemistry, and viral DNA by PCR. Electron microscopy was not performed.
His immunosuppression drugs were reduced (tacrolimus 0.08 mg/kg/day, MMF 1.5 g/day, and prednisolone 10 mg/day). Hematuria settled in a week. His creatinine returned back to 1.0 mg/dl on follow-up (day 52). His creatinine at last follow-up in June 2016 (15 months posttransplantation) was 1.1 mg/dl.
Discussion
Human adenoviruses are a large group of DNA viruses, which cause self-limited respiratory, gastrointestinal, and conjunctival disease in immunocompetent patients. They may be responsible for severe, protracted, and even life-threatening infections in renal and other organ transplant recipients, causing a major impact on morbidity, mortality, and graft survival.[1]
Adenoviruses are classified into seven subgroups, which are further divided into 52 serotypes. The serotypes have different organ tropism. Serotypes 11, 35, and 37 cause renal allograft dysfunction.[1] Infection caused by adenovirus can be classified as (i) asymptomatic adenovirus infection when the virus is detected in urine, blood, stool, or upper airway specimens by viral culture, antigen tests, or PCR, but signs and symptoms of the infection are absent and (ii) adenovirus disease, like our patient, when there are organ-specific signs and symptoms with simultaneous detection of the virus in biopsy specimens by immunohistochemical stains, or from bronchioalveolar lavage or cerebrospinal fluid by culture or PCR, in the absence of another disease. The disease is said to be disseminated when two or more organs are involved, not including viremia.[2]
The incidence of adenovirus infection ranges from 3% to 47% in stem cell transplant recipients and from 5% to 22% in solid organ transplant recipients. In kidney transplant recipients, the reported incidence is 4.1%.[345] Majority of adenovirus infection in transplant recipients is a result of reactivation of a latent infection. It may rarely be due to a primary infection or transmitted through donor organs.[6]
Adenovirus infection is commonly reported early after transplant when the immunosuppression is intense.[7] The mean time of presentation is 3 (1–8) months. Our patient presented on the 23rd posttransplant day, which is uncommon. Late-onset disease, ranging from 17 to 144 months[3] and also causing obstructive uropathy, has been reported.[8]
Hemorrhagic cystitis is the most common clinical manifestation of the infection in renal transplant recipients.[29] Other manifestations of urinary tract involvement include interstitial nephritis, acute tubular necrosis, and ureteral obstruction with hydronephrosis or rarely as a mass lesion in the kidney.[2810] Literature review showed that majority of patients present with gross hematuria, dysuria, fever, and acute graft dysfunction.[1011] Our patient did not have fever or dysuria at any point during this disease.
The different approaches available for the specific diagnosis of adenovirus infection include viral culture, molecular methods, and histopathology. Serologic studies are not commonly used as it has low sensitivity and its significance is uncertain in immunocompromised patients as they may not be able to elicit an immune response.[1]
Many adenovirus serotypes can be isolated in cell culture lines that are commonly used in diagnostic virology laboratories but a few, such as types 40 and 41, fail to grow.[1] Viral culture has only a limited clinical significance in renal transplant recipients as adenovirus can be shed in the urine for a prolonged period in an asymptomatic individual and a positive result may not signify adenoviral disease. Furthermore, it may take up to 28 days for the result.[12]
PCR methods that amplify and detect adenovirus DNA are the commonly used laboratory method to confirm infection with an adenovirus. It is a highly sensitive diagnostic test that gives rapid results. PCR also has the advantage of picking up all adenovirus serotypes and qualitative as well as quantitative assays are available. However, the PCR results should be correlated with the clinical findings to distinguish adenoviral disease from asymptomatic shedding of the virus. Although urine PCR was positive in our patient, the renal biopsy diagnosis confirmed the diagnosis. Serial quantitative PCR is useful for planning the treatment as well as to gauge the response to therapy.[313]
A renal biopsy is very useful in renal transplant recipients to identify histological changes of viral infection, rejection, or other concomitant pathology. The histopathological features include (i) viral inclusion bodies in the nuclei of tubular epithelial cells, (ii) severe tubular destruction with neutrophilic infiltration, cell necrosis, and rupture of tubular basement membranes, (iii) focal necrotizing tubulointerstitial nephritis which may show small granulomas, and (iv) interstitial hemorrhage and intratubular red blood cell casts. Glomeruli and blood vessels are usually spared though very rarely the parietal epithelial cells lining the Bowman's capsule may reveal viral cytopathic changes.[141516] Our patient had the typical viral cytopathic changes along with severe tubular destruction and interstitial hemorrhage on the graft biopsy, but no granulomatous inflammatory response was identified.
The presence of adenovirus within the renal tissue can be confirmed using immunoperoxidase staining and/or in situ hybridization.[141516] The diagnosis in our patient was confirmed by the positive immunoperoxidase staining for adenovirus antigens.
Electron microscopy can identify the typical 70–80 nm diameter adenoviral particles within the nuclei and cytoplasm of tubular epithelial cells. Adenoviruses are usually double the size of BKV and half of that of CMV.[16]
The common differential diagnoses for ADVIN when viral inclusions are identified include BK and CMV nephritis. The presence of interstitial hemorrhage and severe necrotizing lesions disrupting the tubular basement membranes favors adenoviral nephritis.[141516] If a granulomatous inflammation is also present, tuberculosis, sarcoidosis, fungal infections, drugs, and antineutrophil cytoplasmic autoantibody-associated vasculitis need to be ruled out.[16]
Rarely, ADVIN and cellular rejection may coexist and pose a diagnostic challenge. In such situations, the presence of overriding tubulitis, intimal arteritis, and a positive C4d staining on the peritubular capillaries would favor the presence of rejection.[2] There was no glomerulitis or vasculitis in our patient. His C4d immunostain was also negative and so a diagnosis of acute antibody-mediated rejection was ruled out.
Reduction of immunosuppression is the mainstay of treatment. IV immunoglobulin, cidofovir, and ribavirin have been reported as therapies.[39101117] However, no prospective randomized clinical trials have been performed to support the use of any antiviral agent for adenovirus infection.[2] Our patient's graft function improved dramatically just by lowering the dose of immunosuppressants. He was not given any antiviral drug except for the continuation of the CMV prophylaxis.
In summary, our case highlights that adenovirus infection should also be considered in the workup for early graft dysfunction in renal transplant recipients. Awareness of the presenting features and, characteristic biopsy findings supported by immunohistochemical stains and molecular studies help establish a correct diagnosis. This case also highlights that resolution of the infection was achieved by reduction in the immunosuppression.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- What do we know about adenovirus in renal transplantation? Nephrol Dial Transplant. 2013;28:2003-10.
- [Google Scholar]
- Adenovirus disease after kidney transplantation: Course of infection and outcome in relation to blood viral load and immune recovery. Am J Transplant. 2011;11:1308-14.
- [Google Scholar]
- Adenovirus infection in adult orthotopic liver transplant recipients: Incidence and clinical significance. J Infect Dis. 1998;177:459-62.
- [Google Scholar]
- Reactivation of viruses in solid organ transplant patients receiving cytomegalovirus prophylaxis. Transplantation. 2006;82(2 Suppl):S9-14.
- [Google Scholar]
- The impact of adenovirus infection on the immunocompromised host. Rev Med Virol. 2003;13:155-71.
- [Google Scholar]
- AST Infectious Diseases Community of Practice. Adenovirus in solid organ transplantation. Am J Transplant. 2013;13:206-11.
- [Google Scholar]
- Adenovirus nephritis and obstructive uropathy in a renal transplant recipient: Case report and literature review. NDT Plus. 2010;3:388-92.
- [Google Scholar]
- Hemorrhagic adenovirus cystitis after renal transplantation. Transplant Proc. 2004;36:3025-7.
- [Google Scholar]
- Adenovirus in kidney transplantation: An emerging pathogen? Transplantation. 2004;77:1474-5.
- [Google Scholar]
- Subacute allograft failure with dysuria and hematuria in a kidney transplant recipient. Am J Kidney Dis. 2009;54:154-8.
- [Google Scholar]
- Adenovirus infections in stem cell transplant recipients: Recent developments in understanding of pathogenesis, diagnosis and management. Leuk Lymphoma. 2004;45:873-85.
- [Google Scholar]
- Real-time blood plasma polymerase chain reaction for management of disseminated adenovirus infection. Clin Infect Dis. 2004;38:45-52.
- [Google Scholar]
- Necrotizing tubulointerstitial nephritis associated with adenovirus infection. Hum Pathol. 1991;22:1225-31.
- [Google Scholar]
- AST Infectious Diseases Community of Practice. Adenovirus in solid organ transplant recipients. Am J Transplant. 2009;9(Suppl 4):S161-5.
- [Google Scholar]







