Translate this page into:
Myoglobinuria-induced Acute Kidney Injury Secondary to CovishieldTM Vaccination
-
Received: ,
Accepted: ,
This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.
This article was originally published by Wolters Kluwer - Medknow and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Vaccination is the best strategy for the development of herd immunity and for the control of the SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2) pandemic. As the number of immunizations across the globe reaches a record number, random cases of diverse adverse effects of the vaccines are being documented. We report a case of renal biopsy–proven myoglobin-induced acute tubular injury requiring dialytic support post-CovishieldTM vaccination. Awareness of this rare complication is necessary so that it can be recognized early, and renal injury avoided.
Keywords
COVID
myoglobinuria
vaccination
Introduction
The coronavirus disease 2019 (COVID-19) vaccination program in India commenced on January 16, 2021, with two main vaccines, namely, CovishieldTM (ChAdOx1), a recombinant replication-deficient chimpanzee adenoviral vector ChAdOx1 containing the SARS-CoV-2 spike glycoprotein, manufactured under technology transfer from Oxford/AstraZeneca, and Covaxin (BBV152), which is an inactivated whole virion.
The common side effects of ChAdOx1 vaccination reported in the interim analysis include fever, chills, headache, abdominal pain, enlarged lymph nodes, and rash.[1] More recently, a severe vaccine-induced immune thrombotic thrombocytopenia due to anti-platelet factor 4/heparin antibodies has been described in a small postvaccine cohort in two landmark publications,[23] and earlier trials reported three cases of acute transverse myelitis.[4] We describe a case of myoglobinuria-induced acute kidney injury (AKI) occurring postvaccination. The study was approved by the Ethics Committee of the organization vide letter number IEC/PIMS/2021/003.
Case Report
A 47-year-old previously healthy male, with no prior COVID illness, received the first dose of CovishieldTM vaccine, following which he developed generalized weakness and low-grade fever on the third day, which progressed leading to an inability to walk or get up from the sitting position. Low blood pressure was recorded, and he was given a saline infusion at home. This progressed to nausea, vomiting, anasarca, and anuria by the 14th day when he reported to the hospital. There was no history of pyuria, frothy urine, hematuria, pain in the abdomen, or rash. He was perfectly healthy prior to this illness and was nondiabetic, nonhypertensive, with no history of drug intake, alcohol, substance abuse, or smoking. There was no history of the patient having undertaken any strenuous activity or exercise since the day of vaccination. He was a small-time businessman and owned an optical shop. His body mass index was 29.
The investigations done at the time of admission are listed in Tables 1 and 2. The provisional clinical diagnosis was an acute tubulointerstitial nephritis with a rapidly progressive renal failure. He was treated with intravenous (IV) diuretics, pulse doses of 500 mg IV methylprednisolone for 3 days followed by an oral dose of 0.5 mg/kg body weight/day, and he also received five sessions of hemodialysis. A renal biopsy was done on the seventh day of admission to ascertain the cause of the kidney injury.
| Test | Result | Test | Result |
|---|---|---|---|
| Hemoglobin | 10.3 g/dL | CRP | 81.5 mg/dL |
| WBC count | 16,800/mm3 | LDH | 1021 U/L |
| N – 50, L – 43, E – 3, M – 4, B – 0 | |||
| Platelets | 240,000/mm3 | Serum phosphorous/calcium | 6.2 mg/dL/10 mg/dL |
| ESR | 100 mm/hour | Viral serology panel | Negative |
| Urine | Sg – 1.015, protein – 1+, RBC – plenty, WBC – 5-6/hpf | Blood pH | 7.3 |
| pO2 | 113 | ||
| pCO2 | 19 | ||
| Bicarbonate | 9.8 | ||
| Blood urea | 181 mg/dL | Alkaline phosphatase | 94 U/L |
| Serum creatinine | 10.9 mg/dL | AST | 110 U/L |
| Serum sodium | 141 meq/L | ALT | 98 U/L |
| Serum potassium | 3.9 meq/L | Serum bilirubin | 0.6 mg/dL |
| Serum chloride | 102 meq/L | Total protein | 6.3 g/dL |
| Albumin | 3.6 g/dL | ||
| Serum uric acid | 5.6 mg/dL | Procalcitonin | 1.14 ng/mL |
| ANA | 1 + (1:100) | C3 | 1.19 g/L |
| Anti-ds-DNA, c-ANCA, p-ANCA, RA, ASO | Negative | C4 | 0.17 g/l |
CRP=C-reactive protein; WBC=white blood cells; LDH=lactate dehydrogenase; N=neutrophils, L=leucocytes, E=eosinophils, M=monocytes, B=basophils; ESR=erythrocyte sedimentation rate; Sg=specific gravity; RBC=red blood cells; hpf=high-power field; pH=acidity; pCO2=partial pressure of carbon dioxide; pO2=partial pressure of oxygen; AST=aspartate aminotransferase; ALT=alanine aminotransferase; ANA=antinuclear antibody; anti-ds-DNA=Anti-double Stranded DNA; c-ANCA=antineutrophil cytoplasmic autoantibody, cytoplasmic; p-ANCA=peripheral antineutrophilcytoplasmic antibodies; RA=rheumatoid arthritis; ASO=antistreptolysin O
| Test | Result |
|---|---|
| ECG | No ischemic changes |
| 2D echo cardiogram | Normal ventricular function, no regional wall abnormality. No clot or pericardial effusion |
| CT abdomen | Right kidney – 125×69 mm, left kidney – 120×80 mm |
| Blood culture | No growth |
| Urine culture | No growth |
ECG=electrocardiogram, 2D=two-dimensional, CT=computed tomography
The biopsy was adequate with 20 glomeruli and four arteries. All the glomeruli were viable and normal with no increase in cellularity, segmental lesions, or crescents. There was significant tubular injury with many tubules having hyaline and granular casts, and few casts appeared pigmented. The interstitium was widened and edematous with few lymphocytes and occasional plasma cells. There was no significant glomerular obsolescence or tubular atrophy. The blood vessels were normal. Myoglobin immune stains showed strong positivity in the pigmented casts confirming a diagnosis of myoglobinuria-induced acute tubular injury [Figure 1]. Immunofluorescence study showed six glomeruli with absent immune deposits.
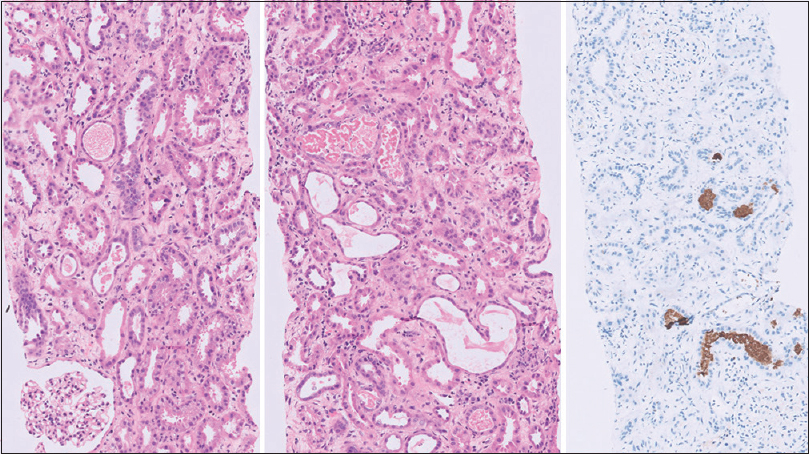
- The left panel shows one normal glomerulus. The tubules show granular casts with focal dilatation and tubular injury (hematoxylin and eosin [HE] 200×). The middle panel shows tubules with granular pigmented casts. The interstitium is widened and edematous with occasional lymphocytes (HE 200×). The right panel shows myoglobin positive casts (IHC myoglobin 100×)
Dialysis was withdrawn after 10 days. The steroids were tapered over 21 days. The steady improvement in creatinine and urine output during the 10 days of admission, from the 15th to the 24th day of vaccination and after discharge till the 76th day are documented in Table 3. The serum creatinine returned to normal at 0.8 mg/dL and blood urea at 19 mg/dL on Day 42 after vaccination, and at the last follow-up on Day 76, the patient continued to be normal. The creatine phosphokinase (CPK) level taken 28 days after the vaccination was normal at 38 U/L (normal <171 U/l). The COVID immunoglobulin G (IgG; neutralizing antibodies) was >20 by chemiluminescence assay at 42 days postvaccination (reactive is >1.0).
| Day postvaccine | Serum creatinine (mg/dL) | Blood urea (mg/dL) | Fluid intake (mL) | Urine output (mL) | Remarks |
|---|---|---|---|---|---|
| 14th | 12 | 300 | 100 | 200 | |
| 15th | 11.2 | 300 | 700 | 900 | Dialyzed |
| 16th | 9.8 | 280 | 2,150 | 1,250 | |
| 17th | 8.8 | 174 | 2,095 | 1,005 | Dialyzed |
| 18th | 10.0 | 189 | 2,015 | 1,420 | |
| 19th | 7.8 | 300 | 900 | 1,290 | Dialyzed, biopsied. |
| 20th | 1,000 | 3,900 | |||
| 21st | 5.4 | 253 | 1,600 | 2,100 | Dialyzed |
| 22nd | 5.8 | 257 | |||
| 23rd | 5.0 | 162 | 1,300 | 1,570 | Dialyzed |
| 25th | 4.0 | 156 | 1,450 | 1,800 | |
| 30th | 2.0 | 85 | |||
| 42nd | 0.8 | 19 | |||
| 48th | 1.0 | 24 | |||
| 76th | 0.8 | 25 |
Discussion
This apparently healthy male with no comorbidity had myoglobinuria-induced acute tubular injury, secondary to rhabdomyolysis. All known causes of rhabdomyolysis were excluded, including trauma, injury, strenuous exercise, alcohol or illicit drug abuse, use of statins, and infections. During the hospital stay, as rhabdomyolysis-mediated kidney injury was not suspected and it is possible that the clinical signs and symptoms may have been masked due to oliguric renal failure, the first CPK level was done nearly a month after the injury, and this was not raised. Although direct causation by the vaccine cannot be concluded, there is a temporal association of the vaccine with the myoglobinuric acute kidney injury.
Vaccine-induced immunological mediated muscle injury is postulated in this case based on the history, the temporal correlation with the vaccine, the elevation of C-reactive protein (CRP), lactate dehydrogenase, SGOT (glutamic-oxalacetic transaminase), SGPT (glutamic-pyruvic transaminase) at the time of admission, the negative procalcitonin values, and the exclusion of other factors mentioned above.
Vaccination induces the production of polyclonal antibodies and T-cell activation (desired immune response) and acute phase reactants such as CRP, interleukin 1 (IL-1), IL-6 (undesired immune response), which may cause negative effects on the recipient.[5] Molecular mimicry and adjuvant-related injury influenced by genetic predisposition are the other possible explanations given for postvaccine immune-mediated injury, including rhabdomyolysis.[6]
There have been a few reports of rhabdomyolysis post COVID vaccination. The vaccine adverse event reporting system maintained by the Centers for Disease Control and Prevention shows 35 events of vaccination-associated rhabdomyolysis as of April 24, 2021. Almost all these are reports of the event after mRNA (messenger ribonucleic acid) vaccines. Myalgias, inability to walk, and elevated creatine kinase have been recorded in the majority. There are, however, no records of renal failure or renal biopsy data in this registry.[7]
One case report of rhabdomyolysis following the Oxford/AstraZeneca vaccine describes a patient with carnitine palmitoyl transferase II deficiency, Day 3 postvaccination with myalgia, weakness, and hematuria and a creatine kinase value of 250,000 U/L.[8] Our patient had no past history to suggest an underlying fatty acid oxidation metabolic disorder, and his liver function tests on follow-up were completely normal. Rhabdomyolysis postvaccination had been reported following influenza,[9] and herpes zoster vaccination,[10] with these patients being on statins at the time of vaccination.
Postvaccination kidney diseases reported are membranous nephropathy linked to influenza, smallpox, minimal change disease to influenza, hepatitis B, polio, measles, DPT (diphtheria–tetanus–pertussis), and pauci-immune glomerulonephritis to influenza vaccine.[11] There are case reports of minimal change disease,[12] relapse of membranous nephropathy,[13] and activation of IgA nephropathy,[14] post COVID vaccination. Immune-mediated injury has been the postulated mechanism in these cases, and we presume that this was so in our case as well.
In conclusion, although the benefits of the vaccine far exceed the rare side effects, we need to be aware of them. This case presents an association of the vaccine with myoglobinuria-induced acute kidney injury. In a patient presenting with unusual body pains or persisting myalgia after the vaccine, investigating for rhabdomyolysis is essential. Our patient had received only one dose of the vaccine, and it is also our opinion that a second dose is best avoided for him.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Oxford COVID vaccine trial group. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: An interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet. 2021;9:99-111.
- [Google Scholar]
- Thrombotic thrombocytopenia after ChAdOx1 nCov-19 vaccination. N Engl J Med. 2021;384:2092-2101.
- [Google Scholar]
- Thrombosis and thrombocytopenia after ChAdOx1 nCoV-19 vaccination. N Engl J Med. 2021;384:2124-30.
- [Google Scholar]
- Acute transverse myelitis (ATM): Clinical review of 43 patients with COVID-19-associated ATM and 3 post-vaccination ATM serious adverse events with the ChAdOx1 nCoV-19 vaccine (AZD1222) Front Immunol. 2021;26:653786.
- [Google Scholar]
- Types of acute phase reactants and their importance in vaccination. Biomed Rep. 2020;12:143-52.
- [Google Scholar]
- Vaccination and autoimmunity-'vaccinosis': A dangerous liaison? J Autoimmun. 2000;14:1-10.
- [Google Scholar]
- Vaccine Adverse Effects Reporting System (VAERS) 1990-5/16/2021, CDC WONDER On-line Database. 2021. https//wonders.cdc.gov/vaers
- [Google Scholar]
- Carnitine palmitoyltransferase II deficiency and post-COVID vaccination rhabdomyolysis. QJM 2021:hcab077. Online ahead of print
- [CrossRef] [Google Scholar]
- Rhabdomyolysis secondary to influenza A H1N1 vaccine resulting in acute kidney injury. Travel Med Infect Dis. 2013;11:130-3.
- [Google Scholar]
- Rhabdomyolysis after recombinant zoster vaccination: A rare adverse reaction. J Community Hosp Intern Med Perspect. 2021;11:145-6.
- [Google Scholar]
- Vaccine-associated kidney diseases: A narrative review of the literature. Saudi J Kidney Dis Transpl. 2019;5:1002-9.
- [Google Scholar]
- Minimal Change Disease Following the Pfizer-BioNTech COVID-19 Vaccine. Am J Kidney Dis. 2021;78:142-5.
- [Google Scholar]
- Relapse of primary membranous nephropathy after inactivated SARS-CoV-2 virus vaccination. Kidney Int. 2021;100:464-5.
- [Google Scholar]
- Gross hematuria following vaccination for severe acute respiratory syndrome coronavirus 2 in 2 patients with IgA nephropathy. Kidney Int. 2021;99:1487.
- [Google Scholar]







